Explication of Saltwater Penetration Behavior and the Evaluation Method
- Seal
- Saltwater intrusion
- Penetration distance estimation
- Evaluation technology
- Electronic equipment
Many electronic devices such as relays, switches and FA sensors are used in the various environments, and they are protected by seal to guarantee their movements. In particular, in applications where outdoor use, water including salt (saltwater) such as seawater and sweat of an operator is attached to the electronic devices. By penetrating the resin housing and dried, if it precipitated as salt crystals in the devices, there is a risk that the conductors are corroded and leading to malfunction.
In this paper, we elucidated penetration behavior of saltwater into the resin housing and precipitation of the crystals. Further, using penetration distance estimation method of the saltwater, it was confirmed that can predict the saltwater penetration of the sealing portion.
It's possible to secure saltwater sealing properties by using obtained knowledge in the design stage and offer the relieved safe electronic devices which can achieve a longer life and reduction of life variations to a customer.
1. Introduction
Most of the electronic devices, such as relays, switches, and FA sensors, are used in the environment of various types and are sealed to ensure proper operation in such environments. One of the purposes of the seal is to ensure electrical insulation and because of such purposes, resins are commonly used. However, the resins are generally water-absorptive and when the electronic device is exposed to water for a long time, water penetrates into the resin housing and short circuits may occur. But the water penetrated into the housing vaporizes when dried and the resin housing returns to the original dry condition.
The electronic devices used outdoors are exposed to saltwater like seawater or sweat of an operator for a long time, and intrusion of saltwater is a concern. Saltwater is a water in which NaCl is dissolved, and when the seawater penetrated in the resin housing dries, NaCl crystalizes in the resin housing when the concentration reaches and exceeds the saturation condition (approx. 26 wt%)1), which is quite a different phenomenon from pure water. When saltwater penetrates into the electronic devices, corrosion and/or short circuiting may occur, and when NaCl crystalizes, continuity of the circuit may be lost by the crystal as the foreign objects. So unexpected failure of an electronic device during operation is of concern if saltwater or NaCl penetrates into an electronic device. However, the mechanisms of saltwater penetrating into the resin housing and where NaCl crystalize by drying are not fully understood.
In the environment of actual use, various factors, such as micro-cracks, due to aging of the resin housing and temperature difference between the inside and outside of the electronic device affect penetration of saltwater, but this paper explains the saltwater penetration behavior in the resin in the simple system of the resin material soaked uniformly in saltwater and then dried and the technique to estimate penetration distance of saltwater and to predict penetration of saltwater to the sealed section developed by the authors.
2. Clarification of Saltwater Penetration Behavior by Immersing and Drying
2.1 Penetration behavior in one cycle of soaking and drying
Sealed construction of the resin housing sealed by the sealant (curable resin like epoxy and urethane resins) as shown Fig. 1 was considered in this study.
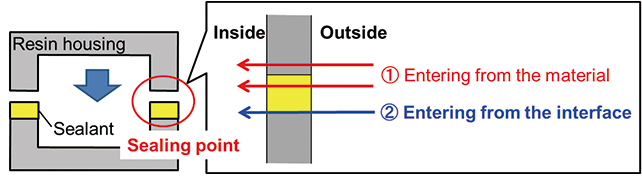
As shown in Fig. 1, saltwater penetrates into the electronic device through the material (resin housing and sealant) and through the interface (joint between the housing and the sealant). As for the interface, OMRON has been successful in reducing the saltwater penetration rate compared to the penetration through the material itself by application of a special treatment. Accordingly, the saltwater penetration rate into the electronic device depends on the penetration rate through the material. Considering the above, the authors studied the saltwater penetration behavior using resin as the material. The material of the resin housing selected was polybutylene terephthalate (PBT), which is commonly used in electronic devises considering insulation ability, thermal resistance, and cost, and material of the sealant selected was epoxy resin used in relays and switches.
The diffusion coefficient can be obtained because liquid penetration into the resin is generally diffusion penetration2) and Fickʼs second law applies. The diffusion distance of saltwater (penetration distance) can be obtained by the general diffusion distance equation3) as shown in Equation (1) using the diffusion coefficient.
-
(1)
Where, 





| Temperature of 5% Saltwater | |||
|---|---|---|---|
| 25℃ | 40℃ | 60℃ | |
| PBT | 3.20×10-7 | 5.55×10-7 | 3.54×10-6 |
| Epoxy Resin | 1.41×10-7 | 3.52×10-7 | 6.45×10-6 |
Table 1 shows that saltwater penetrates more into the resin because the diffusion coefficient becomes larger as temperature increases. The diffusion coefficient of PBT is larger than that of epoxy resin, and saltwater penetrates more into PBT, so it can be considered that PBT controls the rate of saltwater penetration from the material.
Next, in order to verify the certainty of the saltwater penetration distance estimated by the diffusion coefficient, the time required for saltwater to pass through the PBT calculated from Equation (1) and the verification result by the actual material was compared. When the diffusion coefficient at 60°C (3.54±10–6 mm2/s) is used to calculate the time required for saltwater to pass through the PBT of 1.5 mm thick, it is 88 hours.
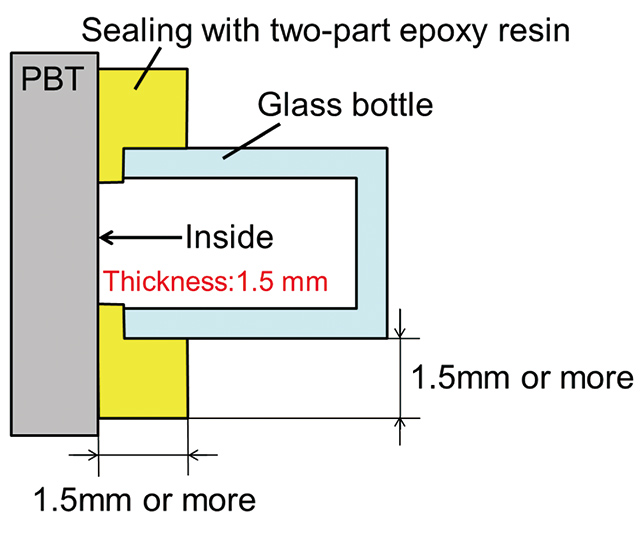
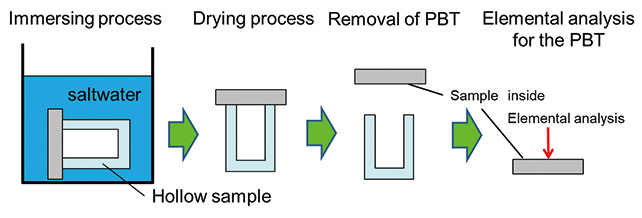
As verification using the actual material, the hollow sample prepared by the PBT sheet and the glass bottle (see Fig. 3) was soaked in saltwater, and the time required for saltwater to pass the PBT sheet of 1.5 mm thick was measured according to the steps shown in Fig. 4. If saltwater passes through the PBT sheet, NaCl will be detected on the inside surface of the PBT sheet where saltwater was not present after the sample is soaked in saltwater and dried. If sodium and/or chlorine element is detected, then it indicates that NaCl is present as crystals of the salt composed of Na and Cl. However, because certain resins like epoxy resin used as sealants may contain chlorine as an impurity, distribution of the sodium element was inspected by the Electron Probe Micro Analyzer (EPMA) in the verification. Although other elemental analysis methods of inorganic compounds like Auger Electron Spectroscopy (AES), X-ray Photoelectron Spectroscopy (XPS), and Secondary Ion Mass Spectrometry (SIMS) are available4), EPMA was used in order to detect quite small amount of sodium element for distribution in the area of a few mm2 area.
Two-part epoxy resin with a smaller diffusion coefficient (penetration of saltwater is difficult) than PBT was used as the adhesive between the PBT sheet and the bottle of the hollow sample in Fig. 3 in order to increase the penetration rate of saltwater with its thickness larger than the thickness of the PBT sheet to prevent penetration of saltwater from the adhesive joint. It is also considered that the internal pressure change due to the increase in the temperature can affect saltwater penetration through the PBT sheet, and in many electronic devices, it is sealed and subject to the effect of internal pressure changes. Accordingly, the interior volume of the bottle was determined so that the interior pressure becomes the same as the pressure that the resin housing of the actual product is subjected to.
The results of the sodium element distribution evaluation by EPMA for the inside surface of the PBT sheet of the hollow sample after 72 and 96 hours of soaking in 5% saltwater at 60°C are shown in Fig. 5. The calculated values of the penetration distance after 72 and 96 hours of soaking were 1.36 and 1.57 mm, respectively.
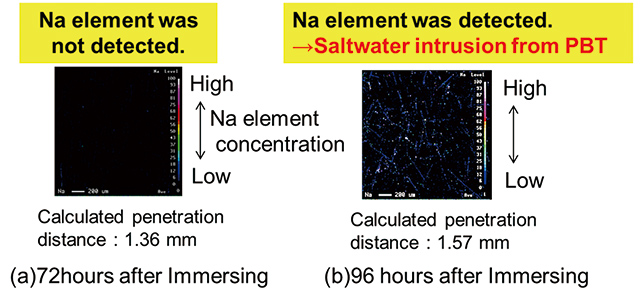
As shown in Fig. 5, sodium element was detected inside the PBT sheet only when the sample was soaked for 96 hours, which indicated deposition of NaCl. So it was confirmed that 72 to 96 hours of soaking is required for saltwater at 60°C to pass through a PBT sheet 1.5 mm thick (calculated value: 88 hours). Thus, it was confirmed that the calculated value is consistent with the verification result of the actual device and penetration distance of saltwater can be estimated from the diffusion coefficient. As sodium element is detected forming lines in Fig. 5, it is considered that NaCl crystalizes mostly on the interface between the glass filler added in PBT and exposed on the surface.
As a failure of the electronic device due to corrosion, shortcircuiting, and loss of continuity of the circuits is likely, when saltwater or NaCl penetrates the device, the presence of saltwater or NaCl in the electronic device is not acceptable.
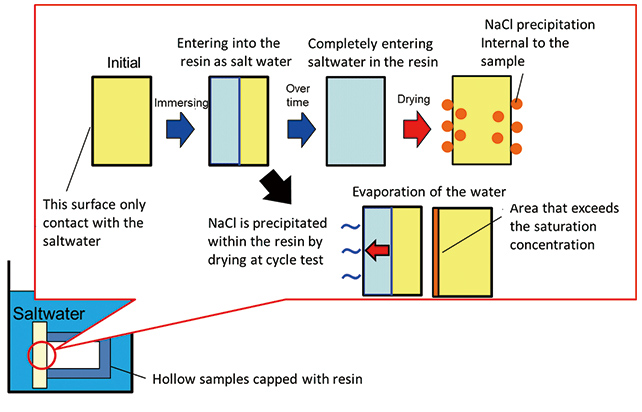
In Fig. 5, it is shown that no deposition of NaCl is observed in the sample after drying, although the calculation shows that the penetration distance after 72 hours of soaking is 1.36 mm, which is as much as 90% of the thickness of 1.5 mm of the PBT sheet. Accordingly, the authors investigated what penetration behavior the saltwater penetrated into the resin will exhibit when soaking and drying cycles are repeated.
2.2 Saltwater penetration behavior caused by soaking and drying cycles
In the soaking process, saltwater penetrates into the resin, and water in the saltwater that penetrated into the resin evaporates in the drying process, and when the concentration of saltwater exceeded the saturation point (about 26 wt.%), NaCl started to crystalize. (For example in the case of 5 wt.% saltwater, the concentration will exceed 26 wt.% when about 81 wt.% of the water vaporizes.) Where the crystalized NaCl from the saltwater penetrated in the resin will be present and whether deposition affects penetration of saltwater are unknown.
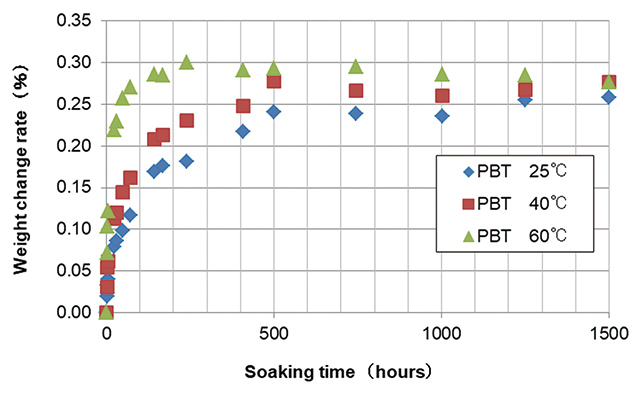
So, the location of NaCl deposition in the resin was investigated using the sample after the test, measuring weight change indicating change of saltwater penetrated in soaking and drying cycle. A resin sample from the bulk (size: 20 mm×20 mm×3 mm) was used as a sample to investigate weight change and deposition position. Polyamide resin one of the engineering plastic with high water absorption capacity was used as the resin sample that has higher water absorption capacity than PBT and exhibits penetration behavior more clearly.
In order to determine the cycle test conditions, the soaking condition where water absorption by the resin sample can be confirmed when it is soaked in demineralized water (weight change ratio: 0.5% or higher) and the drying condition where weight change becomes 0 mg from the soaked condition were investigated. The cycle test conditions were determined as follows based on the results.
<Cycle test conditions>
・Initial drying condition: 100°C×3.5 hrs
・Soaking condition: 40°C×20 hrs
・Drying condition: 100°C×3.5 hrs
・Number of cycles: 10 cycles of soaking to drying after initial drying.
・Saltwater concentration: 5 wt.%
(According to JIS Z 2371: 2015 [Methods of salt spray testing])
The cycle test was conducted in the conditions above, and results of weight change measured are shown in Fig. 6.
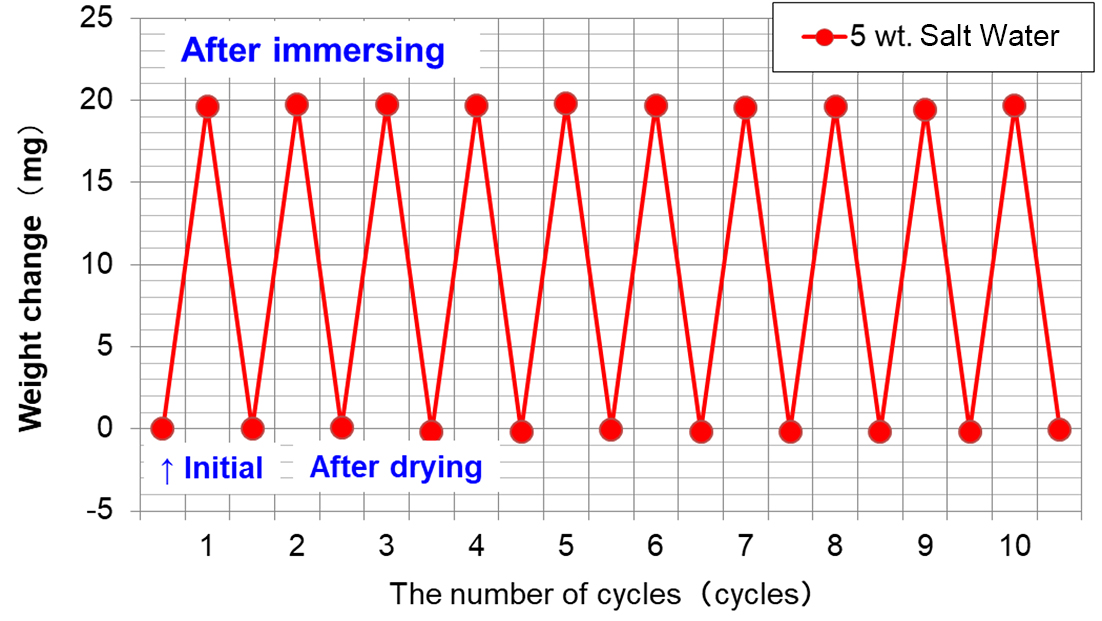
Weight change at the time of soaking does not change significantly even when number of cycles repeated increases as shown in Fig. 6, when the sample is soaked again after drying, amount of saltwater penetrating does not increase, which indicates crystalized NaCl will affect penetration of saltwater a little.
Next, in order to investigate the position where NaCl crystalizes, distribution of the sodium element in the resin was evaluated by line analysis using EPMA. Evaluation result of sodium element distribution in the cross section cut out from the resin sample subjected to 10 cycles of saltwater cycle test is shown in Fig. 7.
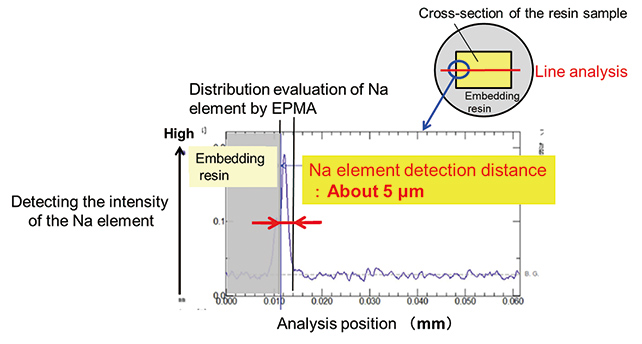
Fig. 7 shows that sodium element was detected to a depth of about 5 μm from the surface of the sample after 10 cycles of soaking and drying, which shows that NaCl crystalized from saltwater penetrated in the resin remains in the resin. In addition to the test results after 10 cycles, Fig. 8 shows the evaluation results of the Na element detection distance for the samples after 5 cycles of test.
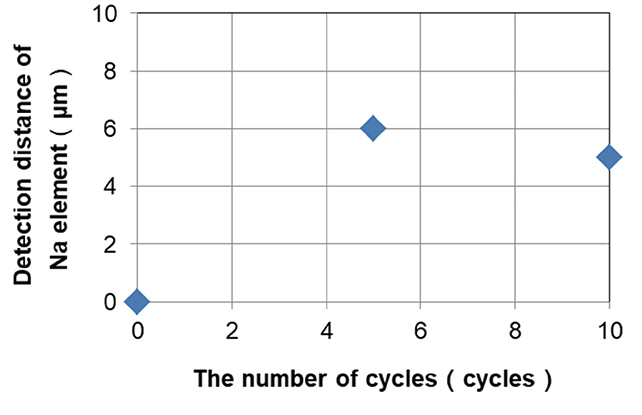
From Fig. 8, it was found that the detection distance of sodium element does not increase with increase of the number of cycles up to 10 cycles, and the detection distance saturates at certain number of cycles.
2.3 Summary of saltwater penetration behavior
Saltwater penetration behavior to the material (resin) obtained from the study can be explained as follows (see Fig. 9). When saltwater penetrates into the resin in the soaking process, the penetration expands into the entire material with time. When saltwater penetrates into the entire resin and is dried, the water content evaporates into the air, the sodium and chlorine ions dissolved in the water migrate to the surface of the resin (both external and internal surface of the hollow specimen), and NaCl crystalizes at the position where saltwater concentration exceeds the saturation concentration. So deposition of NaCl will be found in the resin.
3. Prediction of Saltwater Penetration at the Sealing Portion
3.1 Geometrical concept of sealing portion
Design of the sealing portion is critical to prevent penetration of salt water into the electronic device. The design concept of the sealing portion can be divided to four categories as shown in Fig. 10, when the material and interface that work as the entrance to the saltwater shown in Fig. 1 in Subsection 2.1 and the penetration rate are arranged on the abscissa and ordinate, respectively, when the penetration rate through the resin is used as the basis.
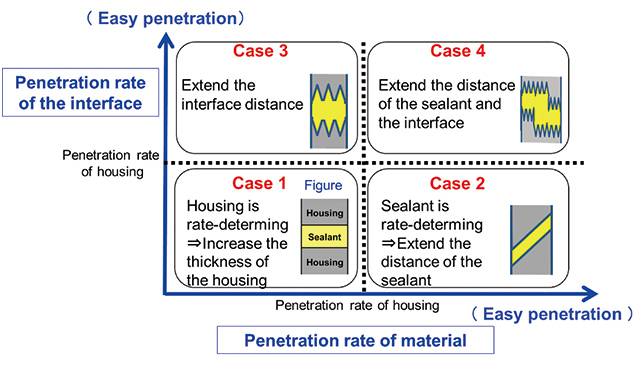
The saltwater penetration rate through the material can be calculated using the diffusion coefficient in Subsection 2.1. OMRON had developed a sealant that has a lower diffusion coefficient than the resin housing material and has lower saltwater penetration rate. As for saltwater penetration through the interface, a surface defect will exist depending on the condition of the interface5), saltwater will penetrate with expansion of the defect (delamination), it is impossible to determine the penetration rate of saltwater using the diffusion coefficient like penetration through the material, and it is anticipated that variability will be large. A large variability is actually confirmed by the test using the actual device. On the contrary, the authors have developed a method to control the penetration rate through the interface by improvement of the surface condition of the resin housing and wettability of the sealant, which made reduction of salt water penetration through the interface possible.
Considering the above, it will be possible to ensure good sealing of saltwater based on the design concept of Case 1 in Fig. 10 with the highest degree of freedom and the least restriction in design by development of the sealant reducing the saltwater penetration rate and by controlling the condition of the interface. When the saltwater penetration test was performed for the specimen with the sealing portion according to Case 1, it was confirmed that saltwater penetration through the resin housing was controllable, which meant that the condition to ensure good saltwater sealing ability was identified.
4. Conclusion
In this paper, the saltwater penetration behavior of the simple system where resin material was uniformly soaked in saltwater and dried repeatedly was explained assuming the electronic devices used in the environment where seawater or the sweat of an operator attached and dried repeatedly. It was found when saltwater penetrated into the resin housing through the resin material, NaCl would crystalize on the electronic device after the seawater dried, which caused a failure due to corrosion, short-circuiting, loss of continuity of the electric circuit. It was also confirmed that salt water penetration behavior could be predicted by selecting appropriate materials for the resin housing and sealant and by controlling the condition of the interface using the saltwater penetration distance estimation method developed in this study.
The knowledge obtained in this study from the design stage will contribute to the supply of safe and secure electronic devices with long service life and reduced variability ensured to the customer, that have good saltwater sealing ability. The authors intend to provide safe and secure products suitable for environment of use to the customer by investigating the penetration behavior of the liquid other than saltwater such as a detergent and cutting fluid and by establishing the method to ensure sealing ability.
References
- 1)
- Chemical Society of Japan. Handbook of Chemistry - Basic Part II. Rev. 3, Maruzen Publishing, 1984, p.173 (in Japanese).
- 2)
- Iwasaki, R.; Yokoyama, N.; Sato, C. Study on the Water Absorption at the Interface and Interphase between Polyurethane Adhesive and Steel Adherend. Journal of the Adhesion Society of Japan. 2007, Vol.43, No.6, p.236-241 (in Japanese).
- 3)
- Kawakami, M. Kinetics of Surface Hardening. Journal of the Metal Finishing Society. 1979, Vol.30, No.4, p.194-199 (in Japanese).
- 4)
- Aono, M. Surface Science Series 5 - Composition Analysis of Surface. Maruzen Publishing, 1999, p.2-3.
- 5)
- Yamazaki, M. Study on Evaluating Internal Interface Strength in Resin-Molded Structures. Doctoral Thesis, Tokyo University, 2010, p.83-88.
The names of products in the text may be trademarks of each company.
















